Key Takeaways
- Scenario: Shift in interventional cardiology toward molecular nanomedicine for treating advanced atherosclerotic lesions.
- Business Impact: Pharma market transition from chronic systemic treatments to high-efficacy targeted therapies reducing residual MACE risk.
- Data Point: Necrotic core area reduction exceeding 30% through CD47 checkpoint inhibitor-mediated efferocytosis restoration.
CD47 Signaling Inhibition: Reprogramming Vascular Immunity
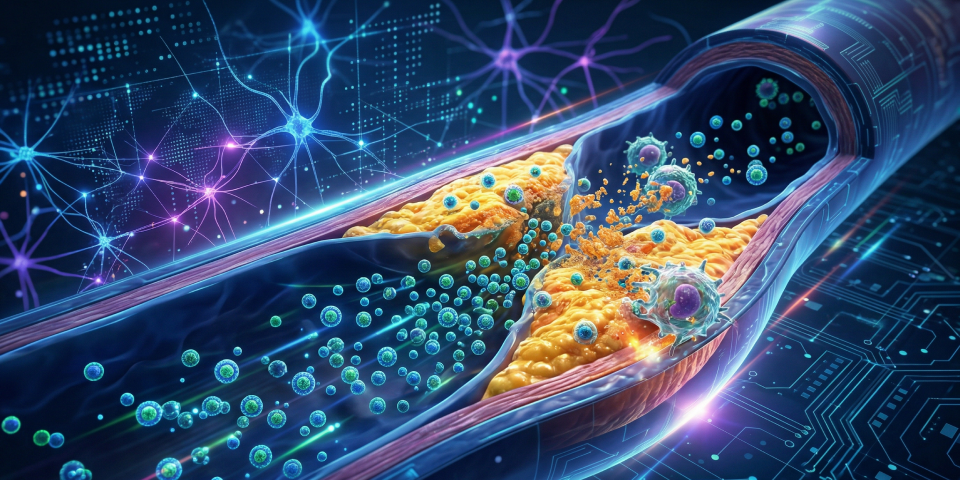
Conventional pharmacological therapies, primarily focused on lowering LDL cholesterol levels, exhibit structural limitations in treating pre-existing plaques at high risk of rupture. Cutting-edge research, featured in Nature Nanotechnology, has identified CD47 signaling (the “don’t eat me” signal) as the primary barrier to natural cellular debris clearance within vessels. Nanomedicina. Apoptotic cells inside the plaque core overexpress this protein, thereby evading macrophage phagocytosis and fueling chronic inflammation.
The utilization of engineered nanoparticles enables the delivery of therapeutic agents, such as SHP-1 phosphatase inhibitors, directly into the vascular microenvironment. Consequently, blocking the CD47-SIRPα axis restores the efferocytosis process, effectively allowing the immune system to “consume” arterial fat and necrotic debris. This biomimetic approach, pioneered by Stanford School of Medicine, minimizes systemic side effects, bypassing the anemia frequently associated with non-targeted anti-CD47 antibodies.
Pharmacokinetics and Selective Targeting of Intra-plaque Macrophages
Treatment precision is ensured by employing single-walled carbon nanotubes (SWNTs) or liposomal carriers that exploit the high permeability of damaged endothelium. These vectors selectively load Ly6C-high macrophages, the primary drivers of plaque instability. However, delivery efficacy does not solely depend on blood concentration; rather, it hinges on the nanocarrier’s capacity to penetrate the deep layers of the tunica intima.
From a bioengineering perspective, the surface functionalization of these nanoparticles ensures prolonged half-life and low immunogenicity. Biotecnologie. This specificity allows for the transformation of a vulnerable “soft” plaque into a stabilized, calcified structure, drastically reducing the probability of distal embolization, a concept supported by clinical insights from the NIH.
Clinical Implications: From Necrotic Core Reduction to MACE Prevention
The success of this technology redefines protocols for the secondary prevention of myocardial infarction. Pre-clinical data indicate that restoring phagocytic activity leads to a significant increase in fibrous cap thickness, a critical parameter for hemodynamic safety. Furthermore, integrating these therapies with advanced diagnostic imaging (PET/MRI) paves the way for theranostics, where lesion identification and treatment occur simultaneously. Technical projections suggest that nanocolloids will become the gold standard for patients resistant to maximal lipid-lowering therapies by 2030.